“Energy cannot be created or destroyed; it can only be changed from one form to another.” This profound insight from Albert Einstein encapsulates the essence of thermodynamics—the science that governs the transformation of energy as we journey from heat to work. In this exploration of thermodynamics, we will uncover the principles that allow us to convert heat energy into useful work, forging a bridge between raw energy and practical applications. This foundational knowledge is not only essential for engineering professionals but also serves as a beacon for students and educators eager to grasp the intricacies of energy interactions.
Delving deeper into the science of thermodynamics will reveal its significance across various fields, including mechanical engineering, chemistry, and environmental science. By comprehending these underlying principles, we pave the way for innovative solutions and advancements in technology. Join us on this enlightening journey as we dissect the laws governing energy transfer and the amazing heat energy conversion.
Key Takeaways
- Thermodynamics governs the conversion of energy from heat to work.
- Understanding thermodynamics is essential for various scientific and engineering fields.
- Albert Einstein’s quote illustrates the fundamental principle of energy conservation.
- Knowledge of thermodynamics inspires innovation in technology.
- Heat energy conversion plays a vital role in real-world applications.
Introduction to Thermodynamics

We are starting an exciting journey into the introduction to thermodynamics. This field is all about energy, showing how heat can turn into work. It has a big impact on many scientific areas. Our first thoughts about heat led to big discoveries and the laws of thermodynamics.
Learning thermodynamics is key for engineers and scientists. It helps in many fields like mechanical, chemical, and environmental engineering. The introduction to thermodynamics teaches us about keeping systems balanced to work better and save energy.
Let’s look at some important ideas in this field:
- The link between heat energy and work.
- The idea of energy staying the same and moving around.
- How it’s used in different engineering areas.
This journey helps us see how thermodynamics has grown over time. It also shows its role in finding green solutions today.
The Science of Thermodynamics
The science of thermodynamics looks into how energy moves in different systems. It’s a key part of physics and many engineering fields. Knowing how energy changes and can be used is very important.
It helps us make things work better, use less waste, and be more efficient.
Understanding Energy Transfer
Energy transfer is a big deal in thermodynamics. It can be heat or work. The laws of thermodynamics say energy can’t be made or lost, only changed.
This idea is why we need to understand how energy moves. It helps us make new and better things in engineering.
Importance in Various Fields
Thermodynamics is very important in engineering. It helps make engines that use less energy. In chemistry, it helps predict how reactions will go and how much energy they’ll use.
Environmental science uses it to study the climate and find ways to use energy better. All these fields need to know about thermodynamics to keep improving.
The Four Laws of Thermodynamics
The laws of thermodynamics explain how energy works in physical systems. Each law has its own role in energy transfer and matter behavior. We have the Zeroth Law, the First Law of conservation, the Second Law of entropy, and the Third Law. They all tell us about energy interactions.
Zeroth Law of Thermodynamics
The zeroth law sets a base for temperature measurement and thermal equilibrium. It says if two systems are in thermal equilibrium with a third, they are in equilibrium with each other. This lets us define temperature scales and compare thermal states.
First Law: Conservation of Energy
The first law says energy can’t be made or destroyed, only changed. The total energy in an isolated system stays the same. This is key for engineering, as it means energy input equals output, helping us design efficient systems.
Second Law: Entropy and Energy
The second law talks about disorder in systems. It says the total entropy of the universe increases in energy transfers. This means energy conversions are never 100% efficient. Knowing this helps us improve processes and design systems that lose less energy.
Third Law: Approaching Absolute Zero
The third law looks at systems as they get close to absolute zero. At this point, all molecular motion stops. The entropy of a perfect crystal gets close to zero. This law shows the limits of energy extraction and helps us understand low-temperature phenomena, leading to advancements in cryogenics and more.
Units and Variables in Thermodynamics

Understanding the units in thermodynamics is key to analyzing systems well. Thermodynamics is built on important thermodynamic variables. These variables affect how systems behave. Some critical ones are:
- Temperature – measured in Kelvin
- Pressure – measured in Pascals
- Volume – measured in cubic meters
- Heat energy – measured in Joules
- Work done – also measured in Joules
Knowing these measurements helps professionals do calculations. It lets them use thermodynamics laws in different ways. Working with the right thermodynamic variables gives us a better understanding of systems. This leads to better decisions in engineering and science.
Understanding Heat and Work

In thermodynamics, heat and work are key to energy transfer and change. Knowing about heat energy helps us make energy systems better and more efficient. This is important in many areas.
Defining Heat Energy
Heat energy is the movement of thermal energy from one system to another because of temperature differences. It moves from warmer to cooler objects until they are the same temperature. This idea is critical for those designing heating and cooling systems.
Work in Thermodynamic Context
In thermodynamics, work is when energy moves an object over a distance because of a force. Examples include gas expanding or compressing. The mix of heat and work is key for improving energy use in systems. It helps in many fields, from mechanical engineering to environmental science.
| Aspect | Heat Energy | Work |
|---|---|---|
| Definition | Transfer of thermal energy due to temperature difference | Energy transfer through force acting over distance |
| Direction | From hot to cold | Can be in any direction based on system design |
| Measurement Units | Joules (J) | Joules (J) |
| Key Example | Heating systems | Gas engines |
Thermodynamic Systems and Their Types

In thermodynamics, we divide systems into three main types: open, closed, and isolated. These thermodynamic systems help us understand how energy and matter move in different settings.
An open system lets both matter and energy flow in and out. For example, a boiling pot of water releases steam. This shows how heat and water vapor leave the system, while the water stays inside.
A closed system can exchange energy but not matter. Think of a sealed container of gas. Heat can move in or out, but the gas itself doesn’t leave the container.
Isolated systems are even more restrictive. They allow neither matter nor energy to leave. A perfectly insulated thermos bottle is a good example. It’s designed to keep heat in or out, without any loss or gain.
Knowing these types of systems is key to understanding and predicting real-world outcomes. Changes in one part can affect the whole system. This influences how the system works and behaves.
From Heat to Work: Exploring the Science of Thermodynamics

The change from heat to work is truly captivating in thermodynamics. Heat engines are key in this process, turning thermal energy into mechanical energy. By understanding their efficiency, we see how these engines work under thermodynamic laws. This knowledge helps us in many real-world uses.
Heat Engines and Their Efficiency
Heat engines work by changing heat into work through cycles. Their efficiency is key, showing how much thermal energy is turned into work. Engines like steam, internal combustion, and gas turbines use different ways to do this. Things like temperature differences and fuel types affect their efficiency.
Real-World Applications
In the real world, the heat to work change is very important. It’s used in power plants to make electricity and in buildings for comfort. Refrigeration systems also use this principle to cool spaces. These examples show our efforts to use energy wisely and reduce waste.
Processes and Cycles in Thermodynamics
Thermodynamics is filled with different processes that show how energy moves and changes. Specific types like isobaric, isometric, and adiabatic processes each have their own way of how systems change. Knowing these processes is key for engineers to make energy systems better, leading to new ideas and more efficiency.
Isobaric, Isometric, and Adiabatic Processes
In thermodynamics, processes are grouped based on their conditions:
- Isobaric Processes: These happen at a constant pressure, letting volume change while heat is exchanged. Seen in heat engines, they help systems do work.
- Isometric Processes: Also called isochoric processes, these keep volume constant. No work is done because volume doesn’t change, letting heat change temperature without changing volume.
- Adiabatic Processes: Here, there’s no heat exchange with the outside. Systems only do work, making it efficient for compressing or expanding gases without losing heat.
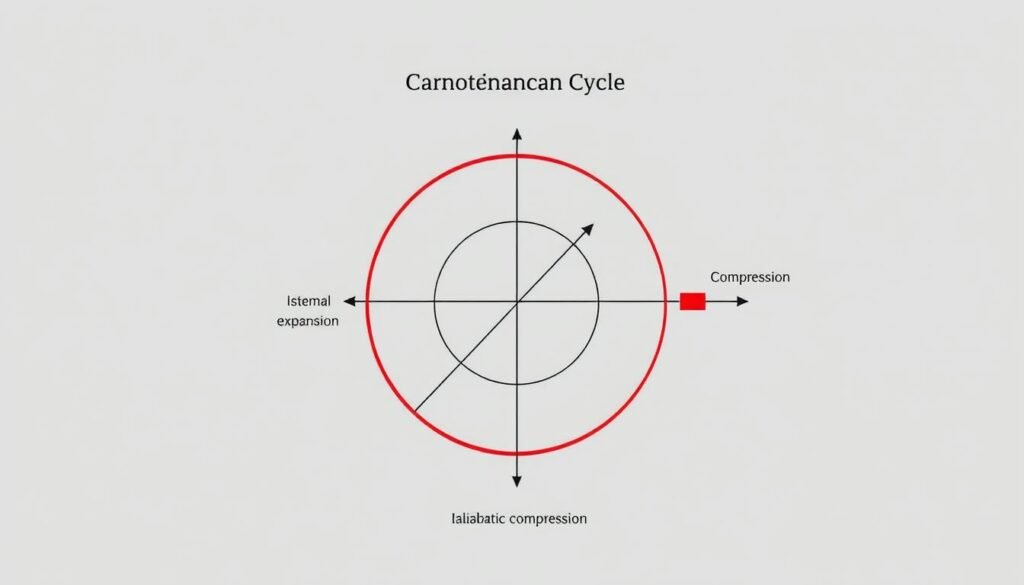
The Carnot Cycle
The Carnot cycle is a key part of thermodynamics, showing the best efficiency possible in heat engines. It has four stages: two isothermal and two adiabatic processes. The Carnot cycle’s importance is in its principles, which set the highest efficiency limits based on temperature differences. It helps engineers make real engines better, aiming for the best energy transfer and less waste.
| Process Type | Description | Applications |
|---|---|---|
| Isobaric | Constant pressure while volume changes | Heat engines, refrigerators |
| Isometric | Constant volume with temperature changes | Gas cylinders, sealed containers |
| Adiabatic | No heat exchange, work done on system | Compression of gases, idealized cycles |
| Carnot Cycle | Two isothermal and two adiabatic processes | Benchmark for heat engine efficiency |
Applications of Thermodynamics
Thermodynamics is key in many fields, driving new ideas and methods. It shows us how these ideas work in real life, like in mechanical engineering, chemistry, and environmental science.
Mechanical Engineering
In mechanical engineering, thermodynamics is basic for making engines and improving power systems. Engineers use it to study heat, fluids, and energy use. This affects how well machines work.
For example, car engines use thermodynamics to turn heat into movement. This makes them more efficient.
Chemistry and Biology Applications
Chemistry uses thermodynamics to predict how reactions will go and if they can happen. It helps scientists make new materials and medicines. It also helps in understanding how living things work.
This knowledge leads to new discoveries in biology, like how cells work and how enzymes react.
Environmental Science Insights
Environmental science uses thermodynamics to study climate and energy use. It helps scientists understand how energy moves in nature and how humans affect it. This knowledge helps find ways to use energy better and protect the environment.
| Field | Specific Applications | Impact |
|---|---|---|
| Mechanical Engineering | Engine design, power generation | Enhanced efficiency, performance optimization |
| Chemistry | Reaction prediction, material development | Innovative solutions, improved stability |
| Environmental Science | Climate modeling, energy sustainability | Resource management, reduced environmental impact |
Measuring Thermodynamic Quantities
Accurate measuring thermodynamics is key for good experimentation and system design. Many instruments help with this, each with its own role:
- Thermometers – These devices accurately measure temperature, providing important data in thermodynamic assessments.
- Barometers – Used to measure atmospheric pressure, barometers help analyze how pressure changes affect thermodynamic states.
- Calorimeters – These instruments measure heat transfer, showing how energy is exchanged in thermodynamic processes.
New technology has made measuring thermodynamics better, leading to more precise experiments. High-resolution sensors and data systems let researchers model and control complex thermodynamic systems well. As these tools get better, they open up new chances in fields like engineering and environmental science.
Entropy and Thermodynamic Potentials
Entropy is key in thermodynamics, showing disorder in systems. It shows how natural processes can’t be reversed. This is vital for the Second Law of Thermodynamics.
As systems change, entropy usually goes up. This means things tend to get more disordered over time.
Thermodynamic potentials like internal energy and enthalpy give us more insight. They help us see how energy changes in phase transitions and chemical reactions. By looking at these potentials, we can predict how a system will act under different conditions.
In real-world uses, entropy and thermodynamic potentials help us understand system stability and efficiency. Using these ideas well can lead to better designs and practices. It’s important for students, professionals, and teachers to grasp this relationship in thermodynamics.
Thermodynamics in Everyday Life
Thermodynamics is all around us, affecting many parts of our daily lives. Think about your fridge, which keeps food fresh. It works by moving heat from inside to outside, using thermodynamic rules. This shows how thermodynamics helps us in our kitchens.
Heating systems also use thermodynamics to keep our homes warm. Whether it’s a furnace or radiators, they spread heat around. This shows thermodynamics in action, making our homes cozy.
When we boil water or brew coffee, we use thermodynamics. Water turning into steam shows energy changes. Seeing these thermodynamic uses in simple tasks helps us understand science better.
Sweating is another example of thermodynamics at work. It cools us down by turning sweat into vapor. This shows how our bodies use thermodynamics to stay cool.
Limitations of Classical Thermodynamics
Classical thermodynamics is key in understanding energy systems. But, it has its limits. It works best with large systems, where we can see the big picture. Engineers use it to design power plants and refrigerators that work well.
But, it struggles with the tiny world. It misses the details of how molecules interact. This is a big problem in nanotechnology and biochemistry, where accuracy matters.
Statistical mechanics helps fill these gaps. It looks at the random nature of molecular interactions. By understanding these limits, we can find new ways to improve efficiency and performance.
| Aspect | Classical Thermodynamics | Statistical Mechanics |
|---|---|---|
| Conceptual Focus | Macroscopic systems | Microscopic systems |
| Molecular Interactions | Ignored | Included |
| Predictive Capability | Effective for large systems | Effective for small-scale systems |
| Real-world Applications | Power generation, Refrigeration | Nano-engineering, Biochemical reactions |
Knowing the limits of classical thermodynamics opens doors to new ideas. It helps us improve our understanding and use of thermodynamics in engineering and science.
Future Trends and Developments in Thermodynamics
Innovation is leading us to exciting times in thermodynamics. Fields like nanotechnology, quantum computing, and bioengineering are changing the game. They bring new ideas and ways to use energy sustainably.
Emerging Fields and Innovations
Nanoscale systems are changing thermodynamics. Working with materials at the atomic level lets us control heat in new ways. Quantum thermodynamics adds more complexity, making us rethink how energy moves.
Researchers are working hard to use these new ideas. They want to make systems more efficient and find better ways to use energy.
Sustainable Energy Solutions
We need to find ways to protect our planet, and sustainable energy is key. New technologies like thermochemical energy conversion and advanced heat pumps are being developed. They help us use energy better and reduce harm to the environment.
As we move forward, we’ll see more ways to use renewable energy together. This will help us make a smoother shift to sustainable energy.
Common Misconceptions about Thermodynamics
In thermodynamics, many myths confuse us, mainly about energy and heat. One myth is that energy is always used well. But, the first law of thermodynamics says energy can’t be made or lost. It doesn’t mean it’s always used best.
This key idea helps clear up mistakes in thermodynamics. It leads to better use of energy in many areas.
Another big mistake is thinking “cold” is a form of energy. But, cold is just the lack of heat. Understanding this is key to getting thermodynamics right. It helps us talk about heat transfers clearly.
By fixing these mistakes, we can better understand thermodynamics. Knowing the truth helps us innovate and teach better. It makes us better at working with energy systems. It also motivates the next generation of scientists and engineers.


Leave a Reply